Safely Increasing Outpatient Health Care: Yesterday, Governor Newsom recommended a resumption of some types of outpatient medical visits, with appropriate precautions to prevent spread of infection. We know you are working on fine-tuning quality and efficiency of your virtual visits, while working to ensure your offices are safe for visits that need to occur in person. The CMA released a high level summary on reopening physician practices. The PHC Medical Director team gathered together a number of best practices for re-opening clinician practices of all types. Clinician Office COVID Guideline is posted separately in this blog. It includes operational, staff, patient flow, and facilities recommendations. We hope it will be a useful check list clinical and operational leaders working to make their offices safe for employees and patients. Some useful additional advice for optimizing video visits can be found in this NEJM article.
Here is a weekly curation of COVID updates that we thought would be of interest to primary care clinicians, with a special focus on many less-recognized clinical manifestations. We appreciate leads on topics for future updates, as well as comments and feedback!
- PHC Policy Updates Related to COVID
- Home Blood Pressure Test Kits: Virtual care of patients with hypertension includes the ability for the patient to check their blood pressure at home. Blood Pressure Test Kits are PHC benefit, provided with a prescription by community pharmacies (not DME vendors). The prescription can specify the size of the cuff that is in the kit (Medium and Large are the two most common), and should include the diagnosis of “hypertension” on the prescription. You may work with your local pharmacy to see what brands are available and write the prescription for that brand. Currently, any BP cuff costing under $55 is covered (this limit is likely to increase soon). Pharmacies that have questions can consult the PHC website for information; please let us know if they refuse to honor a prescription for a BP cuff, and we can reach out to educate them. More information about coverage of Ambulatory BP monitors and BP monitors that automatically send results to the PCP will be presented in a future update.
- Virtual visits by Physical Therapy, Speech Therapy, Occupational Therapy: PHC has activated special codes normally used by MediCare for coverage of video visits by PT/ST/OT to provide education and observation of patients who do not need an in-person visit. We have educated our therapy network of these changes. Patients needing PT/OT/ST services need a prescription from a referring clinician (no need to use the PHC RAF system).
- Role of Serology Testing in Managing COVID. While PCR/RNA/molecular testing is steadily becoming more available, unapproved serology/antibody tests are making their way into the market in a somewhat chaotic fashion. Jill Taylor of the N.Y. State Department of Health stated in an excellent National Academy of Science presentation yesterday that over 125 new serology/antibody tests (many of questionable accuracy) are now available in the United States, although only 5 had official FDA recognition for emergency use. She states that many rapid serology tests will detect milder coronaviruses that cause cold-like symptoms (low specificity), causing rapid surveillance studies to suggest higher rates of immunity than actually exists. Furthermore, we cannot equate presence of an antibody response to true immunity to re-infection. These factors make antibody testing currently unhelpful for counseling individual patients about immunity or safety to return to work. This may change in the near future.
Seroprevalence studies done in early April in Los Angeles and Santa Clara are good examples. Both estimate that about 4% of adults have antibodies in their blood, about 50-fold greater than the number of confirmed positive COVID tests. While some cross-reactivity with milder coronaviruses may be inflating the number, the bottom line is that the vast majority of the population is still susceptible to SARS-CoV2, even in these two counties with California’s heaviest COVID-19 toll. Current estimates are that 60-70% herd immunity would be needed for COVID infection rates to fall.
- How much PCR testing capacity do we need? The current testing rate in the United States is about 0.4-0.5 PCR tests per 1000 population per day. Dr. Ashish Jha of the Harvard Global Health Institute estimated that about 1.5 to 2.5 PCR tests per 1000 population per day will be needed to provide the robust testing of all symptomatic individuals, their asymptomatic contacts, and certain asymptomatic essential workers to safely allow significant resumption of economic activity. For the PHC counties, this would be a total of about 4000-6000 tests per day. This is possible with the current spectrum of tests available, and far more achievable than the 60-100 PCR tests per 1000 population per day recommended by the Roadmap to Pandemic Resilience report released on April 20.
- Additional study on N95 decontamination suggests less repeat decontamination cycles than studies previously reviewed in this newsletter. This study looked at the ability of N95 masks to filter SARS-CoV2 and evaluated integrity using post-sterilization fit testing. This suggested that dry heat sterilization can only be done twice before the N95 fails a fit test. Of note, this study was not yet peer reviewed and comments posted pointed out some important methodological concerns.
- Communication aid for having difficult conversations about COVID. VitalTalks Tips is a free smart phone app that help with difficult conversations with patients, with several sections devoted to COVID-related situations. I recommend loading it on your smart phone and taking a few minutes to scan through some of the questions/topics that are included.
- Therapeutic updates:
- A well-publicized Randomized control trial of hydroxychloroquine showed increased mortality.
- Trial of remdesivir in macaque monkey model: (summary from UCSF ID newsletter) Researchers developed a macaque model of respiratory SARS-CoV-2 infection that recapitulates important aspects of human infections. Investigators from the NIH studiedthe effect of remdesivir treatment starting near the peak of viral replication. Remdesivir was administered with a loading dose 12 hours after respiratory and ocular inoculation, followed by a daily IV dose for 6 days, similar to protocols for humans. At 12 hours after the initial treatment, the remdesivir-treated monkeys had fewer symptoms and less radiologic evidence of pneumonia compared to the control-treated monkeys, a trend which continued during the 7-day study. The remdesivir-treated monkeys had lower viral loads and titers of infectious virus in the lungs and less damage, but interestingly, drug treatment did not decrease viral shedding in the nose, throat, or rectal swabs. While the Gilead remdesivir clinical trial results are not yet known, this primate model suggests that early treatment of COVID-19 with remdesivir may prevent progression to severe pneumonia and sterilize viral cultures in the upper and lower airways. Upper airway viral shedding was still detectable; however, the significance of PCR detectable virus in the absence of viral growth is not known.
- ACEI/ARB not just safe, but also protective: Early data from China indicating that hypertension was a risk factor for COVID severity, combined with the fact that the ACE2 receptor in the lung led to speculation that use of ACEI or ARB increased risk of COVID complications. However, a new report from China showed actually that ACE or ARB use among hospitalized patients with COVID was associated with increased survival.
7. Clinical Updates: As time passes, more reports are showing the wide spectrum of signs and symptoms associated with COVID. Some of these are contrary to the early information we had about the disease, so they are important to incorporate into our clinical decision making, so we don’t reassure patients infected with SARS-CoV2 that they have something else, leading to spread of infection (something we are concerned is occurring, due to the shortage of tests).
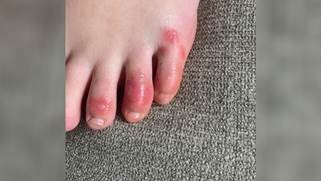
A. Co-infection rate with other respiratory pathogens. A study in Northern California published in JAMA found that 20% of those diagnosed with COVID were co-infected with another respiratory pathogen. Based on this, individuals with symptoms potentially due to COVID should be tested for COVID, even if another test, such as rapid influenza or RSV tests, are positive. B. Smell and taste disturbance in COVID. A physician in Italy noted (in the setting of the epidemic) that sudden anosmia or dysgeusia with nausea –even without other respiratory symptoms—was universally found to be associated with a positive PCR test for COVID. A recent JAMA study of patients with these abnormalities found that most (97%) eventually develop other symptoms of COVID (including even nasal congestion in 35%), but that 12% noted anosmia or dysgeusia as their first symptom. A case report of one such patient found mucous obstruction just below the olfactory nerve, but no imaging abnormality of the olfactory nerve itself, suggesting the etiology may be obstructive, but not ruling out direct infection of the olfactory nerve. C. Why are COVID patients dying at home? The report yesterday that two individuals in Santa Clara who died in early February were found to be infected with COVID is not only interesting for the historical epidemiologic implications, but also as an indicator of an important scary clinical scenario: hypoxia without dyspnea. A NY times article (highly recommended reading) noted that these individuals have pneumonia with decreased oxygen levels but with a normal CO2. They compensate with a more rapid respiratory rate but don’t feel the dyspnea until the pneumonia worsens with elevated CO2, at which time they decompensate quickly, potentially dying at home or requiring intubation by EMS. The article gives anecdotes of the use of oxygen saturation monitors by patients sent home with pneumonia to monitor for detecting hypoxia earlier. PHC is looking into options for making oxygen saturation monitors available to our members. There is a world-wide shortage of these devices currently. D. CT scans identify asymptomatic COVID in New York. Another report from an Emergency Room Physician in New York found that a number of asymptomatic individuals or minimally symptomatic individuals who came to the ED for other reasons were found on CT to have COVID pneumonia. This included a patient with abdominal pain, nausea and vomiting and another with symptoms of acute coronary syndrome. E. Are there cutaneous manifestations of COVID-19?: (summary from UCSF ID newsletter) Cutaneous findings were rarely reported (<1%) inlarge studies from China. A recent study from Italy found that 18 of 88 (20%) hospitalized patients had skin findings: erythematous rash (78%), diffuse urticaria (17%), and vesicles resembling varicella (5%) Another report from Italy described 22 patients with papulovesicular eruption resembling varicella. In both reports, the trunk was most commonly involved, and itching was uncommon. Individual case reports of patients with COVID-19 and a diffuse erythematous rash, diffuse urticaria, petechial rash, and violaceous lesions in the toes have been described as well (sometimes called “COVID toes”), as shown below. Photo: Northwestern University
The American Academy of Dermatology has launched a COVID-19 dermatology registry to better understand the cutaneous manifestations of COVID-19. F. Neurologic manifestations of COVID: (summary from UCSF ID newsletter) What neurological manifestations are seen in patients with COVID-19? Several reports were published this week on neurological findings in patients with COVID-19. A Chinese study described 214 patients and found that 36.4% had neurologic manifestations which included dizziness (16.8%), headache (13.1%), myopathy (10.7%), impaired consciousness (7.5%), taste impairment (5.6%), smell impairment (5.1%), acute stroke (2.8%), and seizure (0.5%). A second French study of 58 ICU patients found agitation (69%), corticospinal tract signs such as clonus (67%), and confusion (65%). 13 patients had MRIs which revealed leptomeningeal enhancement in 62% and acute stroke in 15%. 7 patients had an LP – all were normal and negative for COVID-19 by PCR. Conclusion: Neurologic finding are common in patients with severe COVID-19. Possible mechanisms include hypercoagulability, inflammation, exacerbation of underlying vascular comorbidities, or direct CNS infection. However, only a single case of meningoencephalitis with a positive CSF PCR has been published to date. G. Ocular manifestations of COVID-19: (summary from UCSF ID newsletter) The main ocular manifestation of COVID-19 is conjunctivitis. A recent report described the ocular findings in a cohort of 38 patients from China: 12 patients (38%) had chemosis, conjunctival hyperemia, and epiphora (watery eyes). All 12 patients had moderate, severe, or critical illness, suggesting that ocular findings may be found in more severe disease. Notably, one patient had epiphora (“watery eyes”) as the first symptom of COVID-19. Two out of the 12 patients had a positive conjunctival swab for COVID-19, suggesting the possibility of transmission directly from the eye. Prior to this report, there were also a few case reports where conjunctivitis was described as part of the clinical syndrome of COVID-19. H. Is COVID death sometimes due to multiple blood clots to lungs? (summary from UCSF ID newsletter) Are patients with COVID-19 hypercoagulable and would they benefit from intensified anticoagulation interventions? A report indicates that some patients who die of COVID have many blood clots found in the lungs. A hypercoagulable state is well described in patients with pneumonia and sepsis. Elevated D-dimer and elevated IL-6 (mediator of cytokine induced coagulation) are correlated with poor outcomes in COVID-19. Among 21/183 non-survivors hospitalized with COVID-19 pneumonia, 71% met criteria for disseminated intravascular coagulation. In a retrospective study of severe COVID-19 cases with coagulopathy, heparin was not associated with a benefit in reduction of 28-day mortality except in a subset of patients with very high d-dimers levels. Bottom line: More data are needed to inform these clinical decisions. In the meantime, in light of patient isolation and limited mobility, we agree with The American Society of Hematology (ASH) recommendation that “all hospitalized patients with COVID19 should receive pharmacologic thromboprophylaxis with low molecular weight heparin (LMWH ) or fondaparinux (suggested over unfractionated heparin to reduce contact) unless the patient is judged to be at increased bleeding risk.”
That’s it for this week! We hope this is helpful; again we welcome feedback. We appreciate all that you and your team are doing to support your patients, your community and your families in this challenging time,